Visualizing Biomolecules: X-ray Crystallography

You know you’re a nerd when your tumblr feed lights up with claims like “DNA HELIX PHOTOGRAPHED WITH AN ELECTRON MICROSCOPE FOR THE FIRST TIME!!”. Which is what mine did this past week. We even mentioned this image in the quickies yesterday, linking to an article that blithely repeated the crucial falsehood that it was a single double-stranded DNA molecule. And you know you’re even more of a nerd when you twitch after reading “DNA HELIX PHOTOGRAPHED WITH AN ELECTRON MICROSCOPE!” or “FIRST IMAGE OF DNA!”. So I guess I’m even more of a nerd. I won’t get into the issues with those statements here. Wait, yes I will. But I’ll at least hide most of it below the fold. For someone else’s take, check here.
So, why isn’t this the FIRST PHOTO OF A DNA HELIX!!1? Well, the remarkable image in question (below) isn’t quite a photograph, it’s an electron micrograph. And we’ve taken electron micrographs of DNA before; it’s just generally been chaperoned by proteins. And the strand in the image isn’t a single molecule of DNA, it’s a rope of seven or so molecules (since DNA is double-stranded, that’d be 14 or so strands). So a more accurate sentence might be “First electron micrograph of DNA alone.” Or “First transmission electron micrograph of a naked DNA fiber.” But there’s an even cooler issue here, which is how we know what DNA – or other biomolecules – look like anyway: how do we make pictures of things that are too small to see?

The easy answer would be “We use a microscope”. And while technically correct, light microscopes don’t even get close to the kind of resolution we need to tell what a DNA helix is shaped like. That limitation comes down to wavelength. The human eye can see light between 390 nm and 750 nm in wavelength. A DNA helix is, to be generous, about 2 nm thick and makes a full twist every 3.5 nm or so. Which means that trying to “see” a single, naked, DNA helix with visible light is roughly equivalent to trying to tell what a strand of yarn looks like by throwing a beach ball at it and seeing where the beach ball goes. Unlikely to be very successful.

What we do have, on the other hand, are beautiful pictures of chromosomes taken with traditional light microscopy. At least, when they’re condensed. That’s because a condensed chromosome isn’t just a strand of DNA. It’s a strand of DNA wrapped around histone proteins (in the analogy above, the whole complex would be like a bead on the yarn that was about 6 millimeters in diameter), which are in turned coiled into a rope about 30nm thick (an inch in the beach ball analogy). During mitosis, this rope is looped on itself something like two more times, to make condensed chromosomes somewhere around 700 nm thick (in the analogy: about 20 inches, or two beach balls). Which means, finally, it’s something we could possibly see using visible light. And we do that all the time: a process called karyotyping is often used to screen for serious genetic conditions such as trisomy. Karyotyping is taking a photograph – a real photograph, made by detecting photons – of a cell in metaphase, with all the chromosomes condensed and arranged in rows, and figuring out from that image how many chromosomes the cell has.

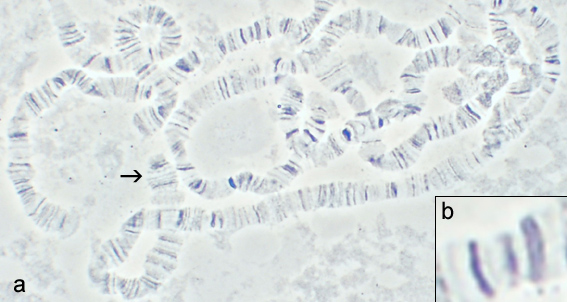
There are certain other times we can use light microscopy to visualize DNA as well. In Drosophila Melanogaster, certain cells have thousands of copies of their genome instead of the usual two. And what’s more, those chromosomes line up in orderly rows. They’re called polytene chromosomes: thousands of strands each 30nm thick. In total, they’re big enough to see with light. And of course, if you purify DNA out of, say, a strawberry, you’ll see a white thread-like goop that “is” DNA (with lots of proteins). It’s all the DNA from the entire strawberry, tangled into a blob. So you certainly can’t tell which part is which. And a lot of the mass is taken up by the proteins that DNA is usually surrounded by. But you can see it.
So, then, how do we have a picture of the 30nm fiber that DNA spends most of its time in? Or, in fact, the 3nm double helix? The answer, at base, is that we use smaller beach balls.
Like I said above, the human eye can’t see light with a wavelength smaller than 390nm or so. That’s violet light. But smaller than violet is ultraviolet, and smaller than that are x-rays. X-rays have a wavelength from 10 to 0.1 nm. So we can throw a bunch of x-rays at something, use a specialized detector to “see” what bounces back, and use that to recreate a picture of a molecule. But wait, I hear a clever person saying: aren’t x-rays the things that pass right through you? If they bounce off of DNA, how can they go through skin, and fat, and organs, and everything in you except bones? And best I can explain is to say that it all depends on targeting and sensitivity. At the doctor’s office, they shoot x-rays indeterminately and most of them pass through soft tissue. They use a fairly sensitive detector that can tell if rays pass through, and that’s how you get the image. So a bunch of the x-rays that they shoot at you can be absorbed, or bounced back in all kinds of directions, and as long as a reasonable portion pass through you still get a good image.
In x-ray crystallography, on the other hand, the beam is tightly focused in on a crystal; an orderly matrix composed entirely (or at least mostly) of the thing to be imaged. In this case, DNA. So that’s difference number one. And the detector is more spread out: we’re looking for the x-rays that get deflected or bent by the crystal, and land “around” the crystal, rather than just those that pass through it (some still may). The point, is, really, that the x-rays that get bounced around and thrown off into space do so in a predictable pattern, and from that pattern we can tell the shape of the molecule that made the crystal. For those of you who love math: we do this with a Fourier transform. For those of you who do not love math: sorry. (As a side note, I think the reason that x-ray diffraction patterns come out as Fourier transforms of images comes down to the same principles as any interference pattern. When you pass a wave through a regular grid – which a crystal will create – you get an interference pattern in two dimensions: a pattern determined by the size, shape, and distance between the cracks between the molecules of the crystal. Said size, shape, and distance between the cracks would be determined by the shape of the molecule itself.)

Why do you have to do all that work, though? When the doctor takes an x-ray to see if you have a broken bone, no Fourier transforms are needed, and crystallization isn’t even possible. The answer again comes down to how the electromagnetic spectrum works. A shorter wavelength means a higher energy. A higher energy means it is more likely to do things like shatter the covalent bonds that hold molecules (like DNA) together. That’s why people worry about x-rays causing cancer: every time you get an x-ray, it probably messes up some of your DNA. So we’re caught with a compromise between short enough wavelength that we can actually “see” a molecule, and low enough energy that we won’t just explode it to bits. Having one molecule sitting there, waiting for its close-up, means you’ll just have a blast-site in the end. Having an orderly grid of millions of copies of that molecule means that, on the average, even if they start degrading as you shoot them with x-rays, the image (the diffraction pattern) stays the same.
One way to get around this problem is to use electrons rather than photons. Electrons are great, because they can have a low wavelength at a (relatively) low energy. And for that reason, electron microscopy is a lot more like light microscopy: you don’t measure a diffraction pattern, you measure an image of the sample. But explaining electron microscopy, and how it works, and why the image I linked above is so remarkable, is a post unto itself.
